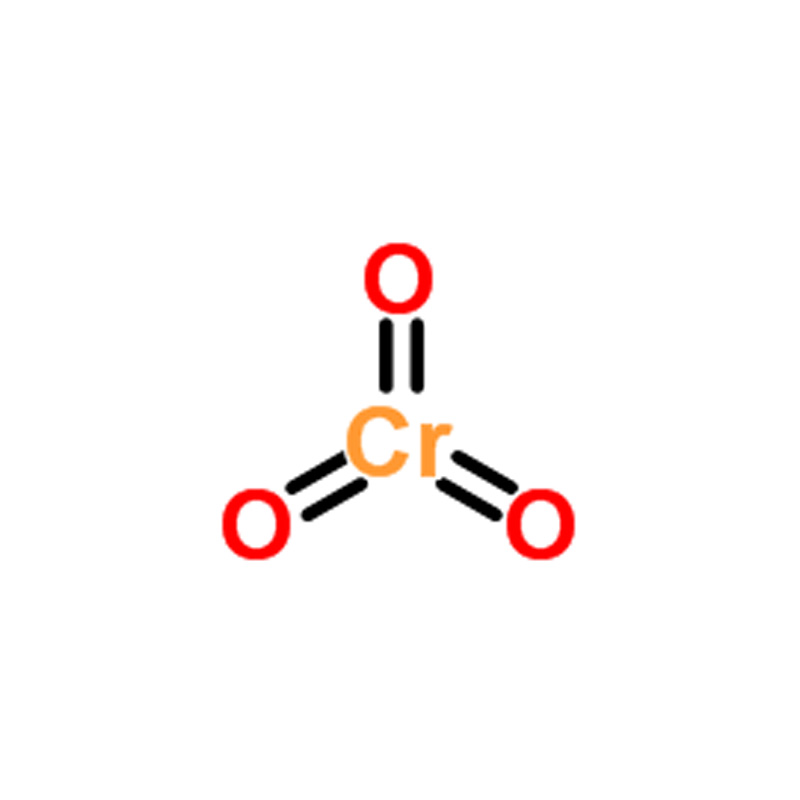
Chromium(VI) oxide CAS#1333-82-0
Exceptional Oxidizing Power
Chromium trioxide is an extremely strong oxidizing agent, highly effective in oxidizing organic and inorganic substances.
High Solubility and Versatile Compatibility
It dissolves readily in water, alcohol, sulfuric acid, and ether, enabling flexible use in various chemical processes and formulations.
Efficient Formation of Chromic Acid
When dissolved in water, it forms chromic acid, a powerful oxidizing and glass-cleaning agent widely used in laboratory and industrial applications.
Thermal Reactivity and Stability
Exhibits defined thermal behavior, decomposing at elevated temperatures (around 400°C) to form chromium(III) oxide, making it suitable for controlled high-temperature applications.
Product Description of Chromium(VI) Oxide CAS#1333-82-0
Chromium(VI) oxide appears as dark red rhombic crystals or purple-red flakes. It is soluble in water, alcohol, sulfuric acid, and ether, but insoluble in acetone. Also known as chromium trioxide (CrO₃), it is a red rhombic compound with a relative density of 2.70 and a melting point of 196°C.
It can be prepared by slowly adding concentrated sulfuric acid to a well-stirred, ice-cooled concentrated aqueous solution of sodium dichromate. The resulting mixture is filtered through sintered glass, washed with nitric acid, and dried at 120°C in a desiccator.
Chromium(VI) oxide is an extremely strong oxidizing agent, particularly toward organic substances, and can immediately ignite ethanol. As an acidic oxide, it dissolves in water to form chromic acid, which is a powerful oxidizing agent and commonly used as a glassware cleaning solution. When heated to about 400°C, it releases oxygen and is converted into chromium(III) oxide.
Chromium(VI) oxide Chemical Properties
| Melting point | 196 °C (dec.)(lit.) |
| Boiling point | 330 °C |
| bulk density | 900kg/m3 |
| density | 2.7 |
| vapor pressure | 0Pa at 25℃ |
| Fp | 250°C |
| storage temp. | Store below +30°C. |
| solubility | 1.667g/l |
| pka | -0.61[at 20 ℃] |
| form | macroporous |
| color | Reddish-violet |
| Specific Gravity | 2.7 |
| PH | <1 (100g/l, H2O, 20℃) |
| Water Solubility | Highly soluble |
| FreezingPoint | 170~172℃ |
| Sensitive | Hygroscopic |
| Merck | 142,235 |
| crystal system | Nogata |
| Space group | Ama2 |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm30.57430.85570.47899090900.2353 |
| Exposure limits | ACGIH: TWA 0.0002 mg/m3; STEL 0.0005 mg/m3 (Skin) |
| OSHA: Ceiling 0.1 mg/m3 | |
| NIOSH: IDLH 15 mg/m3; TWA 0.0002 mg/m3 | |
| Stability: | Stable. Strong oxidizer. Reacts with most organic material in a violent and often explosive fashion. Moisture sensitive. |
| InChI | 1S/Cr.3O |
| InChIKey | WGLPBDUCMAPZCE-UHFFFAOYSA-N |
| SMILES | O=[Cr](=O)=O |
| CAS DataBase Reference | 1333-82-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Chromium trioxide(1333-82-0) |
| EPA Substance Registry System | Chromium(VI) trioxide (1333-82-0) |
Safety Information
| Hazard Codes | O,T+,N,T |
| Risk Statements | 45-46-9-24/25-26-35-42/43-48/23-50/53-62-8 |
| Safety Statements | 53-45-60-61-36/37/39-28-26-22 |
| OEB | E |
| OEL | TWA: 0.0002 mg/m3 (8-hours) |
| RIDADR | UN 1463 5.1/PG 2 |
| WGK Germany | 3 |
| RTECS | GB6650000 |
| TSCA | TSCA listed |
| HazardClass | 5.1 |
| PackingGroup | II |
| HS Code | 28191000 |
| Storage Class | 5.1A - Strongly oxidizing hazardous materials |
| Hazard Classifications | Acute Tox. 2 Inhalation |
| Acute Tox. 3 Dermal | |
| Acute Tox. 3 Oral | |
| Aquatic Acute 1 | |
| Aquatic Chronic 1 | |
| Carc. 1A | |
| Eye Dam. 1 | |
| Muta. 1B | |
| Ox. Sol. 1 | |
| Repr. 2 | |
| Resp. Sens. 1 | |
| Skin Corr. 1A | |
| Skin Sens. 1 | |
| STOT RE 1 Inhalation | |
| STOT SE 3 | |
| Hazardous Substances Data | 1333-82-0(Hazardous Substances Data) |
| Toxicity | LD50 oral (rat) 80 mg/kg |
| PEL (OSHA) 0.1 mg (CrO3)/m3 (ceiling) | |
| TLV-TWA (ACGIH) 0.05 mg (Cr)/m3 | |
| IDLA | 15 mg Cr(VI)/m3 |
Product Application of Chromium(VI) Oxide CAS#1333-82-0
Chromium(VI) oxide is extensively applied in chromium electroplating processes and serves as a protective coating material to enhance the corrosion and oxidation resistance of metal surfaces. It is also utilized in the manufacturing of optical storage media such as CDs and DVDs.
In organic synthesis, it functions as an effective oxidizing agent, particularly in Jones oxidation reactions carried out in acetic acid or acetone. It is commonly used to convert primary alcohols into carboxylic acids and secondary alcohols into ketones.
When combined with phosphoric acid, Chromium(VI) oxide acts as a stripping agent for various anodic coatings and is also involved in the production of synthetic rubies.
Factory and Equipment Show